Product Overview
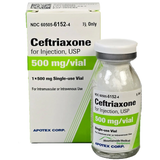
Ceftriaxone for Injection, 500 mg is a sterile injectable formulation supplied in vial presentation for parenteral preparation and administration in healthcare settings. It contains ceftriaxone, a third-generation cephalosporin, and is intended for use within institutional medication preparation workflows.
Product Features
- Supplied as ceftriaxone sterile injectable formulation
- 500 mg strength provided in vial format
- Classified as a third-generation cephalosporin (AHFS)
- Intended for parenteral preparation and administration workflows
- Packaged for use in clinical and institutional healthcare environments
Product Specifications
| SKU/Catalog Number | MF-10180093 |
| Active Ingredient | Cefttriaxone |
| Therapeutic Class | Third Generation Cephalosporin |
| Strength | 500 mg |
| Dosage Form | Powder For Solution |
| Color | White to Yellowish-orange |
| Route of Administration | Intravenous |
| Storage | Controlled Room Temperature (68-77 degrees F) |
| Latex Free | Yes |
| Sugar Free | Yes |
| Dye Free | Yes |
| Alcohol Free | Yes |
| Preservative Free | Yes |
| Hazardous Material | No |