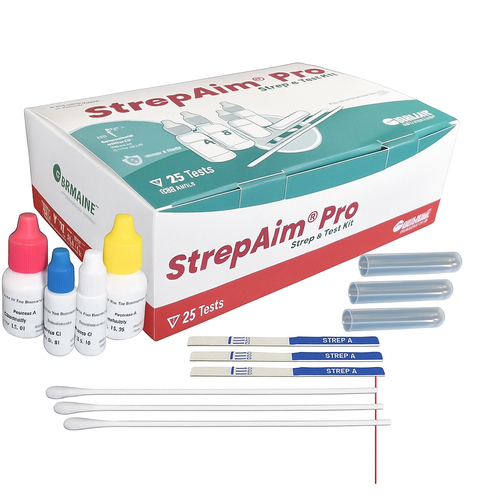
Product Overview
The OSOM Mono Test w/Controls is FDA-cleared with no age restrictions and CLIA-waived for whole blood. It is designed for the qualitative detection of infectious mononucleosis heterophile antibodies in whole blood, serum, or plasma. The new plastic sample transfer pipettes are more secure.
Product Features
- CLIA waived for whole blood.
- FDA cleared
- Sold by the kit
- 25-test set (2 Additional test sticks for external QC)
- Convenient room temperature storage
- Results from a single reagent method can be obtained in 3–8 minutes.
- Fingerstick for whole blood convenience.
- New plastic sample transfer
Product Specifications
| Catalog Number/SKU | MF-SKS-145 |
| CLIA Wave | Yes |
| Test Type | Mono |
| QC Included | 2 Additional test sticks for external QC |
| Rapid Test | Yes |
| Result Time | 3-8 minutes |
| Sensitivity / Specificity | > 99% sensitivity / 96% specificity |
| QTY | 25 Mono Tests per kit |
| Age Limitation | No age limitation |